Tuesday, August 5, 2025
FDA Complete Response Letters: What Actually Blocks Drug Approvals

FDA Complete Response Letters: What Actually Blocks Drug Approvals
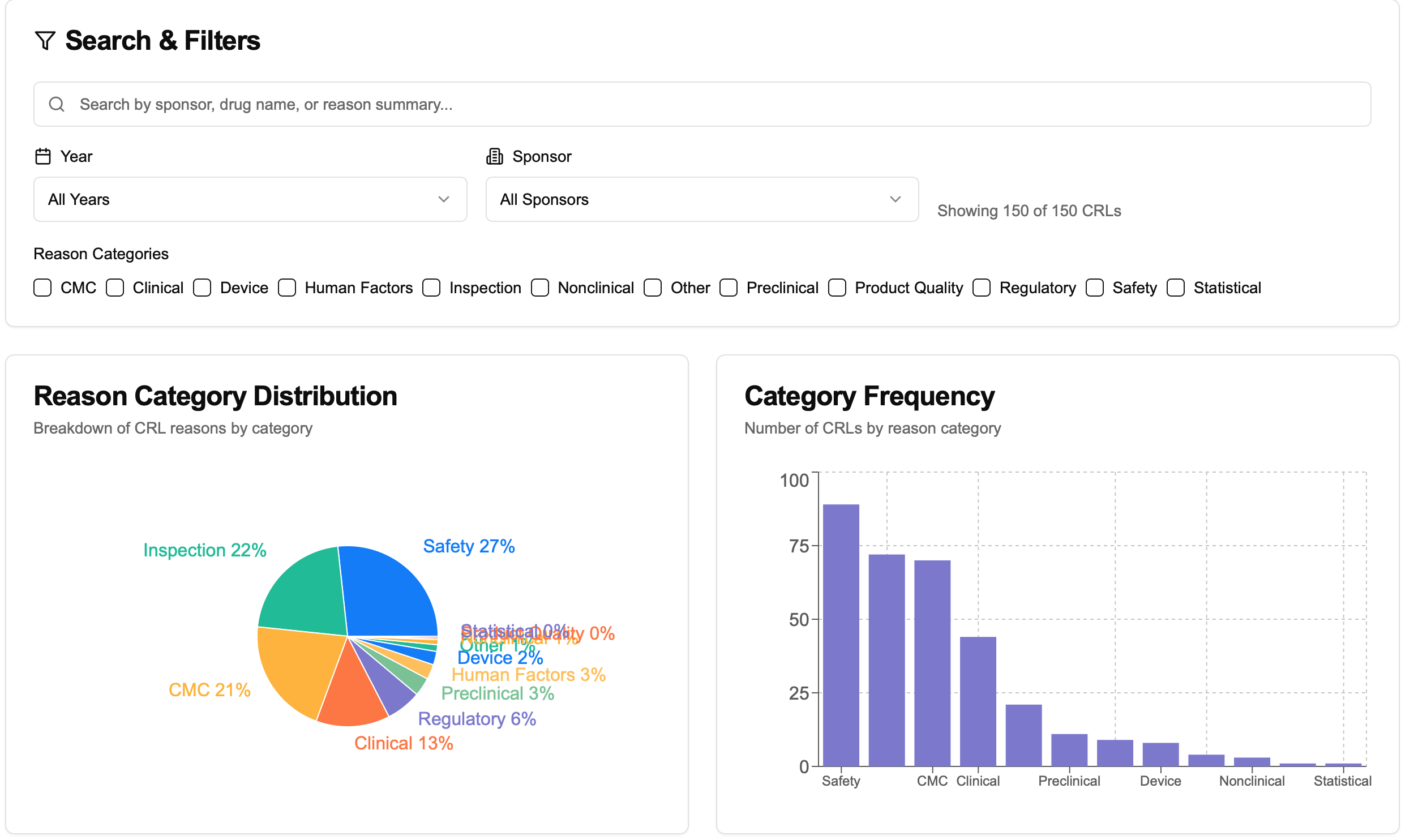
Last week, FDA Commissioner Marty Makary announced that the FDA publicly released nearly 200 Complete Response Letters (CRLs)—formal rejection notices explaining why a drug application could not be approved in its current form. As I was curious about what kinds of issues typically block FDA approvals, I pulled the data via OpenFDA, used AI to extract and classify the rejection reasons, and built a simple dashboard to explore the trends.
🔍 3 Key Patterns That Emerged
Facility inspections (Inspection) were the most common rejection trigger
Frequent GMP violations and unresolved pre-approval inspections, especially in overseas sites.
Many rejections involved combined deficiencies across Clinical, Preclinical, and CMC domains
These weren't single-point failures—the overall submission quality was often insufficient.
Comprehensive safety data updates were widely requested
FDA often required global AE/SAE reports, death cases, and extended exposure data before approval.
What This Means
Actually, these CRLs relate to drugs that were approved later, so there's no need to overanalyze, but still, some of the letters include statistical methods and risk assessment frameworks used by the FDA, making them valuable reference material.
Looking ahead, we may see greater scrutiny around racial diversity in clinical trials, adding new layers to regulatory expectations.
AI Tools in Regulatory Affairs
Meanwhile, AI tools for regulatory use are growing, particularly for narrow, well-defined tasks. As in any medical AI field, compliance, explainability, and transparency remain critical. As the data landscape expands, the choice of model—or hybrid architecture—must be tailored to both the data type and the intended regulatory task.
Global Manufacturing Challenges
Inspection-related issues are frequently cited in CRLs especially in global settings. Does this mean challenges in quality systems, supply chains, and audit readiness across borders? If anyone has experience with this, especially from the CDMO or QA side, I'd love to hear your perspective.
BERSUI Inc. supports corporate productivity improvement and innovative business creation through standardization of AI-driven business operations.
© 2025 Belsui Inc. All rights reserved.

